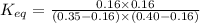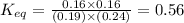In the coal-gasification process, carbon monoxide is converted to carbon dioxide via the following reaction: CO (g) H2O (g) CO2 (g) H2 (g) In an experiment, 0.35 mol of CO and 0.40 mol of H2O were placed in a 1.00-L reaction vessel. At equilibrium, there were 0.19 mol of CO remaining. Keq at the temperature of the experiment is . A) 5.47 B) 1.0 C) 1.78 D) 0.75 E) 0.56

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Which of the following two events occur to create a sea breeze? select all that apply. warm air rises on the ocean and moves toward the land to cool warm air rises on land and moves toward the ocean to cool cool air moves from the ocean to be warmed by the land cool air moves from the land to be warmed by the ocean
Answers: 3

Chemistry, 22.06.2019 05:50
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

Chemistry, 22.06.2019 12:00
Which of the following is an example of physical change not a chemical change? a) a log gives off heat and light as it burns. b) a tree stores energy from the sun in its fruit. c) a penny lost in the grass slowly changes color. d) a water pipe freezes and cracks on a cold night.
Answers: 2

You know the right answer?
In the coal-gasification process, carbon monoxide is converted to carbon dioxide via the following r...
Questions


History, 05.12.2019 03:31



Mathematics, 05.12.2019 03:31


English, 05.12.2019 03:31


English, 05.12.2019 03:31

Business, 05.12.2019 03:31

English, 05.12.2019 03:31


English, 05.12.2019 03:31

Mathematics, 05.12.2019 03:31


History, 05.12.2019 03:31


History, 05.12.2019 03:31


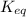 at the temperature of the experiment is 0.56.
at the temperature of the experiment is 0.56. = 0.35 mole
= 0.35 mole
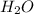 = 0.40 mole
= 0.40 mole
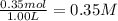
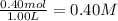
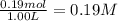
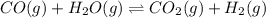
![K_{eq}=\frac{[CO_2]\times [H_2]}{[CO]\times [H_2O]}](/tpl/images/1317/1212/44bdf.png)