
Chemistry, 11.05.2021 08:00 87haymaker
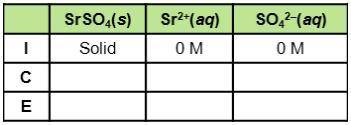
Consider the dissociation of SrSO4, which has a Ksp of 3.2 x 10-7. What do the three rows of (I, C,E) stand for in the table? How can the table be used to find equilibrium constants for this example?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 16:30
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1

Chemistry, 23.06.2019 05:00
Match the term to its description match term definition chemical energy a) internal energy caused by vibrations of atoms and molecules electrical energy b) electromagnetic energy that travels in waves radiant energy c) the movement of an electrical charge thermal energy d) potential energy stored in the bonds between atoms
Answers: 1

Chemistry, 23.06.2019 09:20
Which of the following occurs along coasts during the day?
Answers: 3

Chemistry, 23.06.2019 11:00
Which of the following reactions is endothermic? h2(g) + ½ o2(g) h2o(g), h = -57.82 kcal ½n2(g) + o2(g) + 8.1 kcal no2(g) ½ n2(g) + 3/2 h2(g) nh3(g) + 11.0 kcal c(diamond) + o2(g) co2, h = -94.50 kcal
Answers: 2
You know the right answer?
Consider the dissociation of SrSO4, which has a Ksp of 3.2 x 10-7. What do the three rows of (I, C,E...
Questions

Physics, 10.11.2021 19:40

History, 10.11.2021 19:40

Mathematics, 10.11.2021 19:40


English, 10.11.2021 19:40


History, 10.11.2021 19:40

Mathematics, 10.11.2021 19:40



English, 10.11.2021 19:40

Mathematics, 10.11.2021 19:40

Mathematics, 10.11.2021 19:40

Mathematics, 10.11.2021 19:40


Physics, 10.11.2021 19:40


Mathematics, 10.11.2021 19:40

Mathematics, 10.11.2021 19:40



