
The candle is lit and dilute ethanoic acid is poured down the inside of the beaker. As the acid reacts with the baking soda, bubbles of CO2 gas form. After a few seconds
the air in the beaker is replaced by 0.20 liter of CO2 gas, causing candle flame to go out. The density of CO2 gas is 1.8 grams per liter at room temperature.
Choose the correct structural formula for the acid that was poured into the beaker.
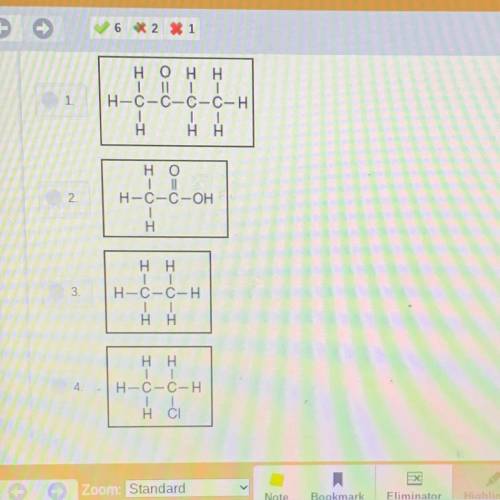

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3
You know the right answer?
The candle is lit and dilute ethanoic acid is poured down the inside of the beaker. As the acid reac...
Questions

Medicine, 24.12.2020 07:30

English, 24.12.2020 07:30


English, 24.12.2020 07:30


Mathematics, 24.12.2020 07:30

Computers and Technology, 24.12.2020 07:30

History, 24.12.2020 07:30

Health, 24.12.2020 07:30

Physics, 24.12.2020 07:30

Mathematics, 24.12.2020 07:30




Mathematics, 24.12.2020 07:30

Mathematics, 24.12.2020 07:30


Arts, 24.12.2020 07:40




