PLEASE HELP!!
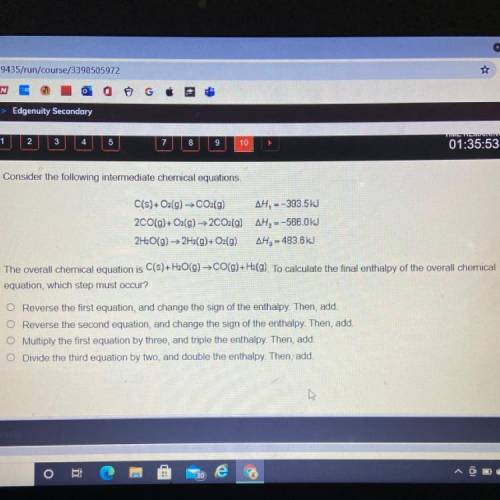
Consider the following intermediate chemical equations.
C(s) + O2(g) → CO2(g)...

Chemistry, 09.05.2021 21:40 jaydahh4059
PLEASE HELP!!
Consider the following intermediate chemical equations.
C(s) + O2(g) → CO2(g) AH, --393.5kJ
2CO(g) + O2(g) → 2CO2(g) AH, --566.0kJ
2H2O(g) → 2H2(g)+O2(g) AH2 - 483.6 kJ
The overall chemical equation is C(s) + H2O(g) → CO(g) +H2(g). To calculate the final enthalpy of the overall chemica
equation, which step must occur?
Reverse the first equation, and change the sign of the enthalpy. Then, add.
O Reverse the second equation, and change the sign of the enthalpy. Then, add.
O Multiply the first equation by three, and triple the enthalpy. Then, add.
O Divide the third equation by two, and double the enthalpy. Then, add.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Select the correct answer. you have a nightlight plugged into an outlet in the hallway, which uses 3.5 watts when plugged in. if the house circuit provides 120.0 volts, what is the current through this bulb?
Answers: 1

Chemistry, 21.06.2019 23:00
The wave shown on the electromagnetic spectrum disturb the medium it passes through a)different frequency. b)the same frequency .
Answers: 2

Chemistry, 22.06.2019 03:40
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3
You know the right answer?
Questions

Mathematics, 16.11.2020 20:40

Chemistry, 16.11.2020 20:40




Physics, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40



Mathematics, 16.11.2020 20:40


Mathematics, 16.11.2020 20:40

Chemistry, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

Physics, 16.11.2020 20:40

Arts, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40




