C(s) +202(g) → CO(9)

Chemistry, 09.05.2021 21:30 YoEsMyles3115
HELPP PLS!
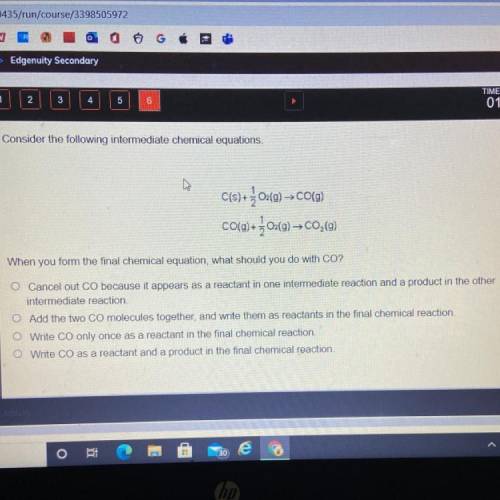
Consider the following intermediate chemical equations.
C(s) +202(g) → CO(9)
CO(g) + 304(9)–C0,(9)
When you form the final chemical equation, what should you do with CO?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 15:00
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2


Chemistry, 23.06.2019 01:00
Reactions in cells take place at about a. 40°c b. 0° c. 100°c d. 60°c
Answers: 1

Chemistry, 23.06.2019 01:00
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1
You know the right answer?
HELPP PLS!
Consider the following intermediate chemical equations.
C(s) +202(g) → CO(9)
C(s) +202(g) → CO(9)
Questions

Mathematics, 25.06.2019 19:20



Geography, 25.06.2019 19:20

Mathematics, 25.06.2019 19:20

Mathematics, 25.06.2019 19:20

Computers and Technology, 25.06.2019 19:20


Mathematics, 25.06.2019 19:20








History, 25.06.2019 19:20

Mathematics, 25.06.2019 19:20


Social Studies, 25.06.2019 19:20



