
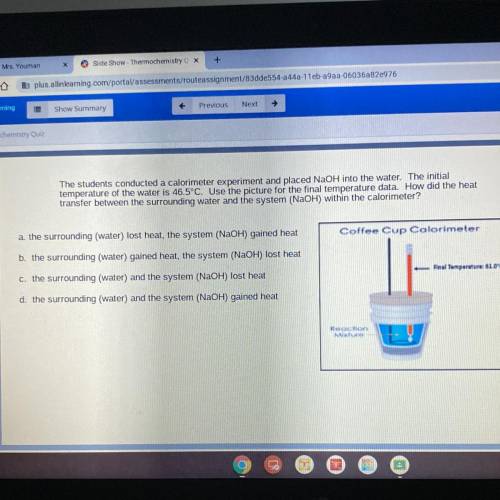
The students conducted a calorimeter experiment and placed NaOH into the water. The initial
temperature of the water is 46.5°C. Use the picture for the final temperature data. How did the heat
transfer between the surrounding water and the system (NaOH) within the calorimeter?
a. the surrounding (water) lost heat, the system (NaOH) gained heat
Coffee Cup Calorimeter
b. the surrounding (water) gained heat, the system (NaOH) lost heat
Final Temperature61.0°C
c. the surrounding (water) and the system (NaOH) lost heat
d. the surrounding (water) and the system (NaOH) gained heat
Reaction
Mixture

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1

Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
The students conducted a calorimeter experiment and placed NaOH into the water. The initial
temper...
Questions

Mathematics, 26.02.2021 07:00

Mathematics, 26.02.2021 07:00

Mathematics, 26.02.2021 07:00

Mathematics, 26.02.2021 07:00


Mathematics, 26.02.2021 07:00


Mathematics, 26.02.2021 07:00


Social Studies, 26.02.2021 07:00

Advanced Placement (AP), 26.02.2021 07:00

Arts, 26.02.2021 07:00


Social Studies, 26.02.2021 07:00



Mathematics, 26.02.2021 07:00

History, 26.02.2021 07:00

Mathematics, 26.02.2021 07:00



