
Chemistry, 08.05.2021 08:10 sparky1234
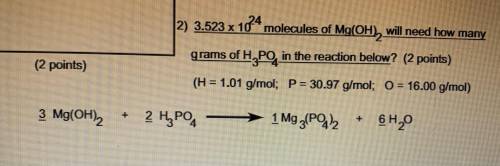
3.523 x 10^24 molecules of Mg(OH2) will need how many grams of H3PO4 in the reaction below
H=1.01 g/mol
P=30.97 g/mol
O=16.00 g/mol
3 Mg(OH)2 + 2 H3PO4 -> 1 Mg3 (PO4 )2 + 6 H2O

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3



Chemistry, 22.06.2019 11:00
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2
You know the right answer?
3.523 x 10^24 molecules of Mg(OH2) will need how many grams of H3PO4 in the reaction below
H=1.01...
Questions

Mathematics, 23.04.2021 16:30




Physics, 23.04.2021 16:30

Mathematics, 23.04.2021 16:30



Chemistry, 23.04.2021 16:30


Mathematics, 23.04.2021 16:30



Mathematics, 23.04.2021 16:40


Mathematics, 23.04.2021 16:40


Mathematics, 23.04.2021 16:40

Health, 23.04.2021 16:40



