
Chemistry, 07.05.2021 20:40 Serenitybella
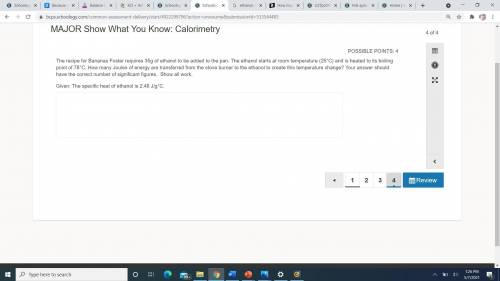
The recipe for Bananas Foster requires 35g of ethanol to be added to the pan. The ethanol starts at room temperature (25°C) and is heated to its boiling point of 78°C. How many Joules of energy are transferred from the stove burner to the ethanol to create this temperature change? Your answer should have the correct number of significant figures. Show all work. Given: The specific heat of ethanol is 2.46 J/g°C.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2

Chemistry, 22.06.2019 15:30
Which of the following are correct values for the ideal gas laws constant r
Answers: 1

You know the right answer?
The recipe for Bananas Foster requires 35g of ethanol to be added to the pan. The ethanol starts at...
Questions

Mathematics, 02.12.2021 17:30


Mathematics, 02.12.2021 17:30

Mathematics, 02.12.2021 17:30

Biology, 02.12.2021 17:30

Mathematics, 02.12.2021 17:30


Geography, 02.12.2021 17:30



History, 02.12.2021 17:30

Business, 02.12.2021 17:30


Mathematics, 02.12.2021 17:30



Spanish, 02.12.2021 17:30

Mathematics, 02.12.2021 17:30





