
1. Calculation of equilibrium concentrations from Ka
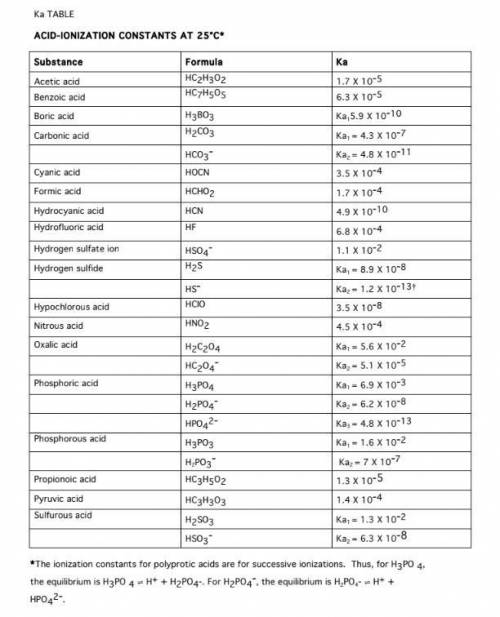
Calculate the pH of a 1.0 M Acetic acid solution, using approximations. Ka = 1.8 x 10^-5
HC2H3O2+H2O⇆H3O(+)+C2H3O2(-)
Initial:
Change
at Equilibrium:
2. Calculation of species concentrations from Ka, using the quadratic formula
Calculate the pH of a 0.000010 M Acetic acid solution
HC2H3O2+H2O⇆H3O(+)+C2H3O2(-)
Initial:
Change:
at Equilibrium:
3. Calculation of Ka from the pH of a weak acid solution
Calculate the Ka of HNO2 if a 0.10 M HNO2 solution has a pH of 2.187
Initial:
Change:
at Equilibrium:
4. Calculation of Ka from the percent ionization
Calculate the Ka of Glycine if a 0.10 M Glycine solution is 4.1 x 10^-3 ionized
HGly+H2O⇆H3O(+)+Gly(-)
Initial:
Change:
at Equilibrium:

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 09:30
In 2002, the rare earth elements mine in mountain pass, california was closed because
Answers: 1

Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 22.06.2019 12:00
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1
You know the right answer?
1. Calculation of equilibrium concentrations from Ka
Calculate the pH of a 1.0 M Acetic acid solut...
Questions


Mathematics, 10.01.2020 03:31


Biology, 10.01.2020 03:31

Mathematics, 10.01.2020 03:31

Mathematics, 10.01.2020 03:31

History, 10.01.2020 03:31



Mathematics, 10.01.2020 03:31


Computers and Technology, 10.01.2020 03:31




Mathematics, 10.01.2020 03:31

Mathematics, 10.01.2020 03:31

English, 10.01.2020 03:31

Mathematics, 10.01.2020 03:31



