
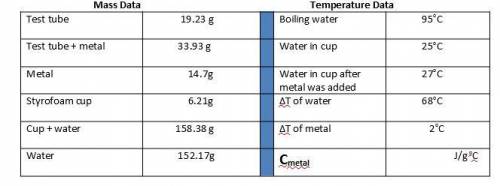
1. Calculate the heat gained by the water (lost by the metal) in the calorimeter using the equation in the introduction.
Metal A
Q = mc(ΔT)
Qwater = -Qmetal
Heat gained = Mass of x Specific heat of x Change in temperature
by the water water (g) water (4.184 J/goC) (ΔT)
The specific heat of the metal can now be calculated:
Specific heat = Heat gained by the water
of metal (c) Mass of metal (g) x ΔT of metal (oC)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 23.06.2019 04:10
Which of the following is described by the equation h2o(s)+ heat=h2o(i) a freezing melting condensing evaporating
Answers: 2
You know the right answer?
1. Calculate the heat gained by the water (lost by the metal) in the calorimeter using the equation...
Questions

Mathematics, 13.11.2020 21:50

History, 13.11.2020 21:50


Mathematics, 13.11.2020 21:50

Mathematics, 13.11.2020 21:50

SAT, 13.11.2020 21:50



Mathematics, 13.11.2020 21:50

Mathematics, 13.11.2020 21:50

Biology, 13.11.2020 21:50

Mathematics, 13.11.2020 21:50

Chemistry, 13.11.2020 21:50

Mathematics, 13.11.2020 21:50

Mathematics, 13.11.2020 21:50

Mathematics, 13.11.2020 21:50


Arts, 13.11.2020 21:50




