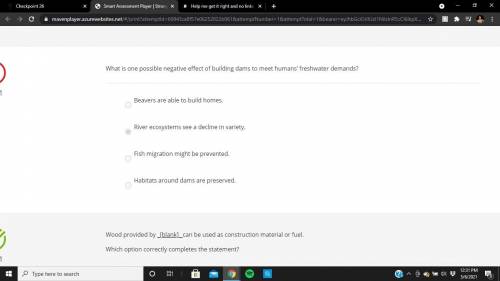
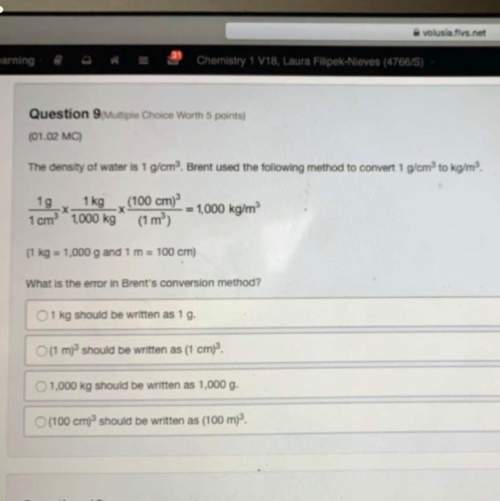
Help me get it right and no links
...

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 22:30
What is the value of the standard enthalpy of formation of an element in its most stable form?
Answers: 3

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 01:30
Use the periodic table to determine how many grams of oxygen would be required to react completely with 859.0 g c2h2
Answers: 3

Chemistry, 23.06.2019 03:00
The size (radius) of an oxygen molecule is about 2.0 ×10−10m. make a rough estimate of the pressure at which the finite volume of the molecules should cause noticeable deviations from ideal-gas behavior at ordinary temperatures (t= 300k ). assume that deviatons would be noticeable when volume of the gas per molecule equals the volume of the molecule itself.
Answers: 3
You know the right answer?
Questions


Mathematics, 07.02.2022 01:00

Mathematics, 07.02.2022 01:00

Mathematics, 07.02.2022 01:00


Mathematics, 07.02.2022 01:00

Biology, 07.02.2022 01:00


Mathematics, 07.02.2022 01:00



Social Studies, 07.02.2022 01:00





Mathematics, 07.02.2022 01:00

English, 07.02.2022 01:00

Mathematics, 07.02.2022 01:00

Health, 07.02.2022 01:00