
Chemistry, 05.05.2021 21:40 jasperzhouzihe3018
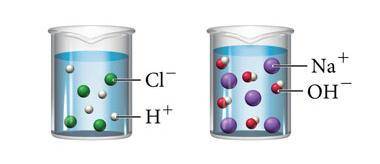
The image above shows what happens when hydrochloric acid (HCl) and sodium hydroxide (NaOH) are mixed with water. Which statement below is true about both solutions?
A. Both solutions contain solutes that do not dissolve completely in water.
B. Both solutions are saturated because they contain the maximum concentration of a solute dissolved in the solvent.
C. Both solutions will conduct an electric current because they both contain ions that can carry a charge.
D. Both solutions are acidic because they both contain hydrogen.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2


Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1
You know the right answer?
The image above shows what happens when hydrochloric acid (HCl) and sodium hydroxide (NaOH) are mixe...
Questions


Mathematics, 19.02.2022 07:10




Mathematics, 19.02.2022 07:10

English, 19.02.2022 07:10



Geography, 19.02.2022 07:10


Mathematics, 19.02.2022 07:20

Computers and Technology, 19.02.2022 07:20

Mathematics, 19.02.2022 07:20



Biology, 19.02.2022 07:20


Mathematics, 19.02.2022 07:20



