
Chemistry, 05.05.2021 02:10 peachijmin
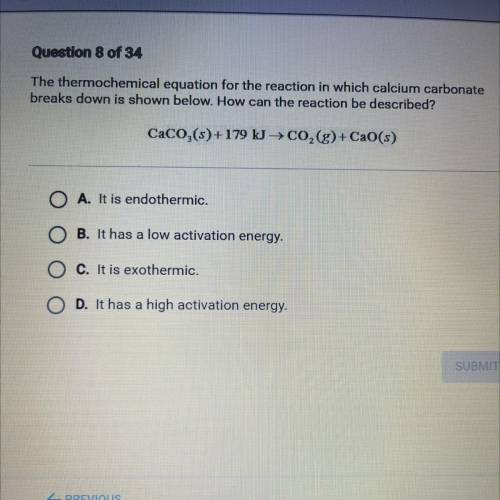
The thermochemical equation for the reaction in which calcium carbonate
breaks down is shown below. How can the reaction be described?
CaCO,(s)+179 kg CO,(g)+ CaO(s)
A. It is endothermic.
B. It has a low activation energy.
C. It is exothermic.
D. It has a high activation energy.
NO LINKS

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
18. use the activity series to predict whether the following synthesis reaction will occur. write the chemical equations for the reaction if it's predicted to occur. (s) + o2(g) -> *note: it is possible.*
Answers: 1


Chemistry, 22.06.2019 11:00
The twister and runaway train are two coasters at the same amusement park. both coasters start at the same height. the coaster for the twister is twice the mass of the coaster for the runaway train. which roller coaster has greater gravitational potential energy at the start of the ride?
Answers: 1

Chemistry, 22.06.2019 17:00
Complete each row of the table below by filling in the missing prefix or missing exponent.
Answers: 1
You know the right answer?
The thermochemical equation for the reaction in which calcium carbonate
breaks down is shown below...
Questions

Mathematics, 19.01.2021 17:40


Mathematics, 19.01.2021 17:40






Mathematics, 19.01.2021 17:40

Mathematics, 19.01.2021 17:40

Mathematics, 19.01.2021 17:40

English, 19.01.2021 17:40

English, 19.01.2021 17:40

Arts, 19.01.2021 17:40


Mathematics, 19.01.2021 17:40

Mathematics, 19.01.2021 17:40


Mathematics, 19.01.2021 17:40

Chemistry, 19.01.2021 17:40



