
Chemistry, 03.05.2021 19:20 ayowazzzgood
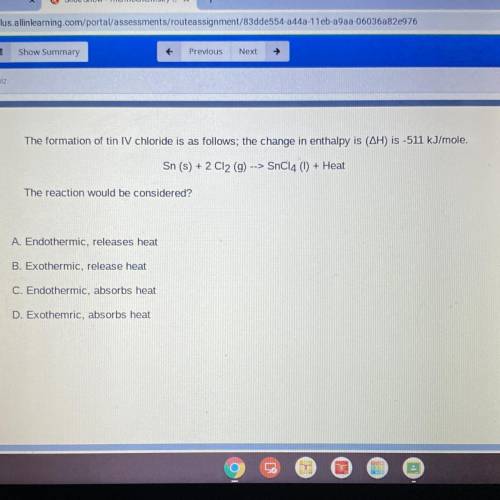
The formation of tin IV chloride is as follows; the change in enthalpy is (AH) is -511 kJ/mole.
Sn (s) + 2 C12 (9) --> SnC14 (1) + Heat
The reaction would be considered?
A. Endothermic, releases heat
B. Exothermic, release heat
C. Endothermic, absorbs heat
D. Exothemric, absorbs heat

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
This active feature of earth's crust in building mountain ranges as well as islands. this feature is a a) cavern. b) earthquake. c) mountain. d) volcano.
Answers: 2

Chemistry, 22.06.2019 04:40
In which environment would primary succession occur? a forest with a few remaining trees after a recent wildfire an area of exposed rock after a glacier melts away beach that is exposed to the air at low tide an abandoned baseball field in a small town
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1
You know the right answer?
The formation of tin IV chloride is as follows; the change in enthalpy is (AH) is -511 kJ/mole.
Sn...
Questions

Mathematics, 01.07.2020 15:01


Mathematics, 01.07.2020 15:01

Mathematics, 01.07.2020 15:01




Medicine, 01.07.2020 15:01


Mathematics, 01.07.2020 15:01

Mathematics, 01.07.2020 15:01


Mathematics, 01.07.2020 15:01


Mathematics, 01.07.2020 15:01


Mathematics, 01.07.2020 15:01



Advanced Placement (AP), 01.07.2020 15:01



