
Chemistry, 03.05.2021 17:10 garrettm162
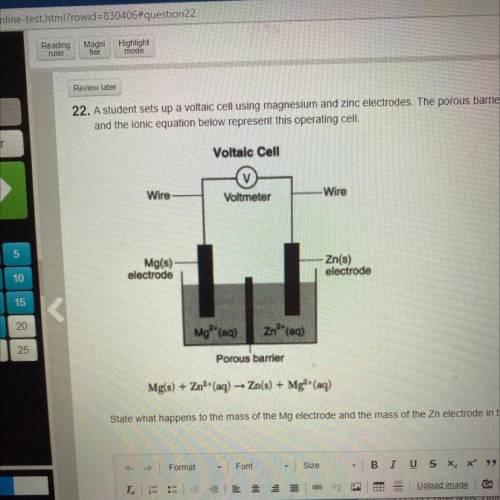
State what happens to the mass of the Mg electrode and the mass of the Zn electrode in the operating cell.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

Chemistry, 22.06.2019 21:00
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1

Chemistry, 22.06.2019 22:10
Which aqueous solution of ki freezes at the lowest temperature? 1) 1 mol of ki in 500. g of water 2) 2 mol of ki in 500. g of water 3) 1 mol of ki in 1000. g of water 4) 2 mol of ki in 1000. g of water
Answers: 3

Chemistry, 23.06.2019 03:00
In which of the following phases of matter do molecules have the highest amount of energy? a. liquid b. gel c. solid d. gas
Answers: 2
You know the right answer?
State what happens to the mass of the Mg electrode and the mass of the Zn electrode in the operating...
Questions


History, 21.01.2021 14:20

Biology, 21.01.2021 14:20



Mathematics, 21.01.2021 14:20


Social Studies, 21.01.2021 14:20

Mathematics, 21.01.2021 14:20

Social Studies, 21.01.2021 14:20


Chemistry, 21.01.2021 14:20

Mathematics, 21.01.2021 14:20

Mathematics, 21.01.2021 14:20

Biology, 21.01.2021 14:20


Mathematics, 21.01.2021 14:20

Mathematics, 21.01.2021 14:20

Mathematics, 21.01.2021 14:20

Mathematics, 21.01.2021 14:20



