
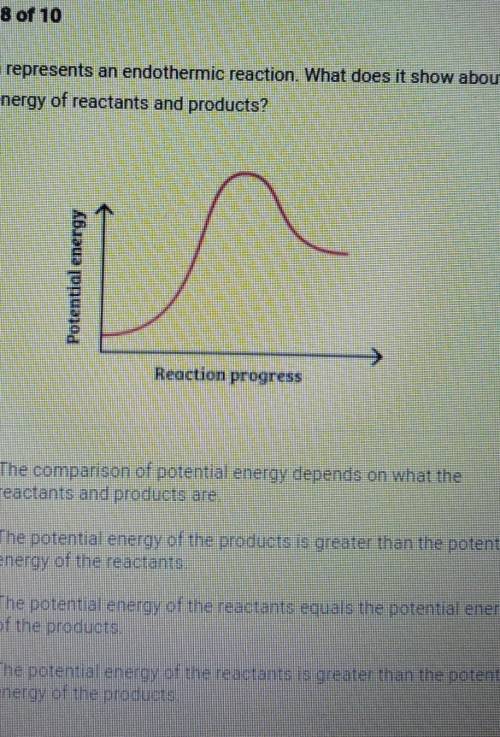
This graph represents an endothermic reaction. What does it show about the potential energy of reactants and products? Abuzua nunuad in Reaction progress A. The comparison of potential energy depends on what the reactants and products are, B. The potential energy of the products is greater than the potential energy of the reactants C. The potential energy of the reactants equals the potential energy of the products D. The potential energy of the reactants is greater than the potential energy of the products,

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:00
If a planet rotates 360 degrees during a 24 hour time period, what does that tell us about the planet? a. the middle of the planet is in darkness b. the seasons on the planet vary every day. c. the planet runs on a 12-hour time clock. d. the temperature on the planet varies daily.
Answers: 1

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2

Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
You know the right answer?
This graph represents an endothermic reaction. What does it show about the potential energy of react...
Questions

Mathematics, 04.05.2021 23:10


Mathematics, 04.05.2021 23:10



Mathematics, 04.05.2021 23:10

Mathematics, 04.05.2021 23:10


Mathematics, 04.05.2021 23:10


Mathematics, 04.05.2021 23:10

Physics, 04.05.2021 23:10


Mathematics, 04.05.2021 23:10


English, 04.05.2021 23:10

Mathematics, 04.05.2021 23:10

English, 04.05.2021 23:10

Business, 04.05.2021 23:10

Social Studies, 04.05.2021 23:10



