
Chemistry, 02.05.2021 23:40 2023brewerantonio
A. Write a series of balanced equations, and fill in the blanks for an overall equation, for the complete oxidation of 2 mol of glutamate to 2 mol of α-ketoglutarate and 1 mol of urea. Note that the overall equation (net reaction) does not include aspartate, fumarate, or oxaloacetate, so you must include balanced equations for the interconversion of these molecules. (Hint: what pathway converts fumarate to oxaloacetate?) For reference, the urea cycle figure is shown on the back. You will need a total of 9 to 11 reactions in order to generate the balanced overall equation.
Overall equation/net reaction (fill in stoichiometry):(The stoichiometry of water and H+is not important here, so don’t stress about that)2 glutamate + __CO2+ 4 H2O + __NAD++ __ATP →2 α-ketoglutarate + urea + ___NADH + 7 H++ __ADP + __AMP + __Pi+ __PPi
b. Based on the final equation above, does the process result in a net gain or loss of ATP equivalents? How many?
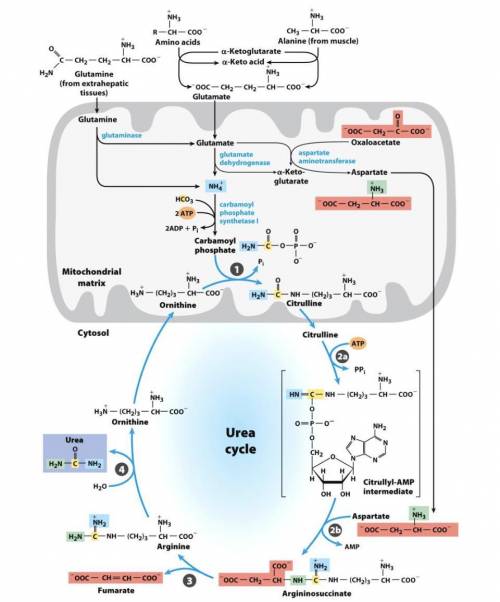

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
What is the force of attraction between the particles in a salt crystal
Answers: 2


Chemistry, 22.06.2019 13:30
Apush or pull that moves or changes and object when to objects touch
Answers: 2

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2
You know the right answer?
A. Write a series of balanced equations, and fill in the blanks for an overall equation, for the com...
Questions



Mathematics, 03.11.2019 08:31




Chemistry, 03.11.2019 08:31

Mathematics, 03.11.2019 08:31

Mathematics, 03.11.2019 08:31

Mathematics, 03.11.2019 08:31

Social Studies, 03.11.2019 08:31

Mathematics, 03.11.2019 08:31







Computers and Technology, 03.11.2019 08:31



