
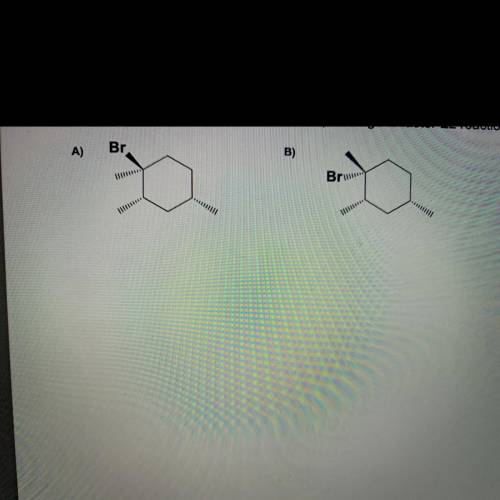
Question 1 (5 points)
The following two compounds A) and B) undergo E2 reaction through B-elimination with sodium
methoxide.
1) Draw the product(s), indicate the major product if any, and give the mechanism to account for the
formation of the product(s) using chair conformation(s) for each compound.
2) Indicate which compound (A or B) undergoes faster E2 reaction and give a brief explanation.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Mrs. smith ordered a root beer float (vanilla ice cream + root beer). mrs. smith noticed that the three states of matter (solid, liquid, and gas) all existed simultaneously in her root beer float. a. identify each phase of matter in the root beer float. b. describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? ) c. identify one phase change you would see in a root beer float and described what causes this change.
Answers: 2

Chemistry, 21.06.2019 21:30
Fugu, also known as puffer fish, is a sushi delicacy that can also be lethal. puffer fish contain a powerful toxin that can kill an adult a few hours after ingestion. sushi chefs who prepare fugu must be specially trained because any contamination of the toxin-free areas of the fish can be deadly. recently this toxin has been put to good use, as scientists have discovered that a purified form of it can treat severe pain in cancer patients. this recent scientific discovery would fall under which area of chemistry? applied biochemistry pure organic chemistry pure physical chemistry applied inorganic chemistry
Answers: 1

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3
You know the right answer?
Question 1 (5 points)
The following two compounds A) and B) undergo E2 reaction through B-eliminat...
Questions

Social Studies, 22.08.2019 15:30



Mathematics, 22.08.2019 15:30

History, 22.08.2019 15:30


Biology, 22.08.2019 15:30

Biology, 22.08.2019 15:30

Chemistry, 22.08.2019 15:30

History, 22.08.2019 15:30


History, 22.08.2019 15:30


Social Studies, 22.08.2019 15:30




Biology, 22.08.2019 15:30

History, 22.08.2019 15:30



