Please
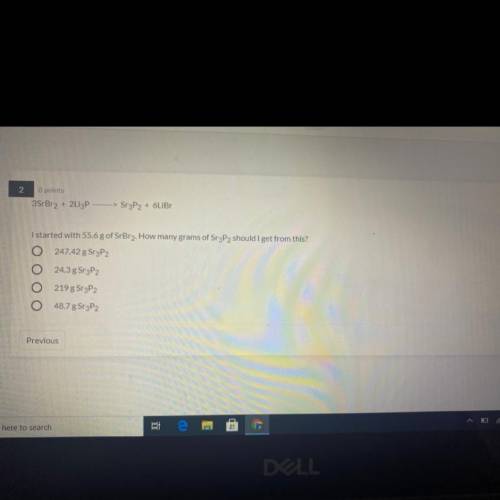
3SrBr2 + 2Li3P —>Sr3P2 + 6LiBr
I started with 55.6 g of SrBr2. How many gram...

Chemistry, 28.04.2021 19:30 rwlockwood1
Please
3SrBr2 + 2Li3P —>Sr3P2 + 6LiBr
I started with 55.6 g of SrBr2. How many grams of Sr3P2 should I get from this?
A) 247.42 g SrP2
B) 24.3 g Sr3P2
C) 219 g Sr3P2
D) 48.7 g Sr3P2

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:00
How many mmols of tris-hcl are there in 100 ml of a 100 mm tris-hcl buffer solution at ph 8.1? note that the 100 mm refers to the sum of tris and tris-hcl concentrations?
Answers: 3

Chemistry, 22.06.2019 11:00
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced.be sure your answer has the correct number of significant digits.
Answers: 2

Chemistry, 22.06.2019 13:30
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1
You know the right answer?
Questions





Social Studies, 30.07.2019 16:00




Biology, 30.07.2019 16:00



History, 30.07.2019 16:00

Social Studies, 30.07.2019 16:00


Mathematics, 30.07.2019 16:00

History, 30.07.2019 16:00

Chemistry, 30.07.2019 16:00



Mathematics, 30.07.2019 16:00



