
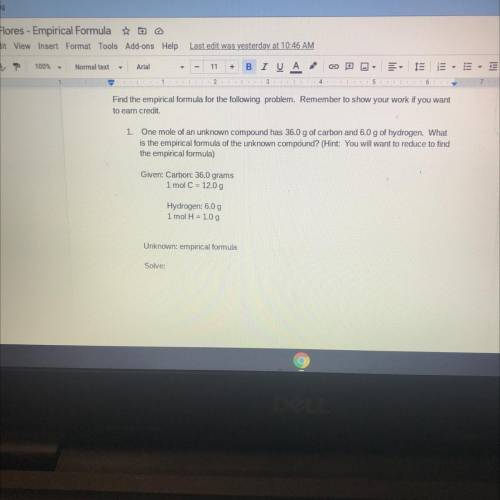
PLEASE HELP DUE IN 20 MINUTES Find the empirical formula for the following problem. Remember to show your work if you want
to earn credit.
1. One mole of an unknown compound has 36.0 g of carbon and 6.0 g of hydrogen. What
is the empirical formula of the unknown compound? (Hint: You will want to reduce to find
the empirical formula)
Given: Carbon: 36.0 grams
1 mol C = 12.0 g
Hydrogen: 6.0 g
1 mol H = 1.0 g
Unknown: empirical formula
Solve:

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 23.06.2019 00:30
Ok, so i have 2 questions. try to answer them both: (the topic is fire) 1) how can you represent the chemical reaction of fire? 2) what kind of bond is formed in this chemical reaction
Answers: 3

Chemistry, 23.06.2019 07:30
If you try to move a piano and are unable to move it, did you perform any work in the scientific sense of the word? yes or no?
Answers: 1
You know the right answer?
PLEASE HELP DUE IN 20 MINUTES Find the empirical formula for the following problem. Remember to show...
Questions


Biology, 28.07.2019 16:00


Mathematics, 28.07.2019 16:10



Mathematics, 28.07.2019 16:10

Mathematics, 28.07.2019 16:10



Social Studies, 28.07.2019 16:10


Computers and Technology, 28.07.2019 16:10


History, 28.07.2019 16:10

Biology, 28.07.2019 16:10

Social Studies, 28.07.2019 16:10


Computers and Technology, 28.07.2019 16:10

Chemistry, 28.07.2019 16:10



