
Chemistry, 27.04.2021 01:00 Cjdjejfjiv8568
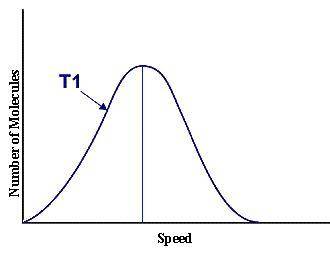
This graph represents a population of molecules in a gas versus the distribution of the average velocity(speed) of its molecules in that population. Assume all molecules to be of the same mass. In reading the graph, it is important to note three things. One, is the most probable speed is at the peak of the curve. Secondly, the most probable speed increases as the temperature increases (so shift to the right), and the distribution broadens as it increases.
On the graph, indicate the average kinetic energy of the population.
Explain your answer.
What part of the graph indicates the temperature of the sample?
Explain your answer.
Print out graph paper (click here for graph paper) and sketch a curve that represents the distribution of molecules at a temperature below the one shown. Label it as T2. Describe both T and T2 in terms of their average kinetic energy. Be specific and detailed.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2


Chemistry, 22.06.2019 23:00
Which type of intermolecular attractions holds ammonia molecules together with other ammonia molecules?
Answers: 3
You know the right answer?
This graph represents a population of molecules in a gas versus the distribution of the average velo...
Questions

Physics, 04.10.2019 23:00

Mathematics, 04.10.2019 23:00

Mathematics, 04.10.2019 23:00



Mathematics, 04.10.2019 23:00

History, 04.10.2019 23:00

Social Studies, 04.10.2019 23:00


Mathematics, 04.10.2019 23:00



Mathematics, 04.10.2019 23:00

Mathematics, 04.10.2019 23:00


Biology, 04.10.2019 23:00



Biology, 04.10.2019 23:00



