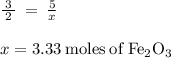Chemistry, 26.04.2021 21:30 roperbailey
Calculate the number of moles of iron (III) oxide (Fe 2 O 3 ) produced from 112 L of oxygen
(O 2 ) in the following reaction
4Fe(s) + 3O2 (g) = 2Fe2O3 (s)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:30
Select each correct answer. more than one answer may be correct. which of the following is a characteristic of unicellular organisms? they can possess tissues and organs. all of their functions are performed by a single cell. they are usually microscopic. each of their cells is specialized to perform a specific function.
Answers: 1


Chemistry, 23.06.2019 03:10
Which of the following compounds would be expected to have the strongest ionic bonds? a)the compound that has b)the largest ions with the greatest charge c)the compound that has d)the largest ions with the least charge the compound that has the smallest ions with the greatest charge the compound that has the smallest ions with the least charge
Answers: 2

Chemistry, 23.06.2019 04:31
How does a sample of helium at 15 degree celsius compare to a sample of helium at 215 k? a) the helium at 15 degrees celsius has a higher average kinetic energy that the sample at 215 k. b) the helium at 15 degrees celsius has lower nuclear energy that the sample at 215 k. c) the helium at 15 degrees celsius has slower- moving atoms that the sample at 215 k. d) the helium at 15 degrees celsius has smaller atoms than the sample at 215 k.
Answers: 1
You know the right answer?
Calculate the number of moles of iron (III) oxide (Fe 2 O 3 ) produced from 112 L of oxygen
(O 2 )...
Questions


History, 11.05.2021 21:40

English, 11.05.2021 21:40

History, 11.05.2021 21:40



Biology, 11.05.2021 21:40


Advanced Placement (AP), 11.05.2021 21:40

History, 11.05.2021 21:40

Mathematics, 11.05.2021 21:40

Mathematics, 11.05.2021 21:40


Mathematics, 11.05.2021 21:40

English, 11.05.2021 21:40

Mathematics, 11.05.2021 21:40



Mathematics, 11.05.2021 21:40

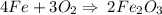

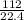 (Since one mole at STP = 22.4 L)
(Since one mole at STP = 22.4 L)