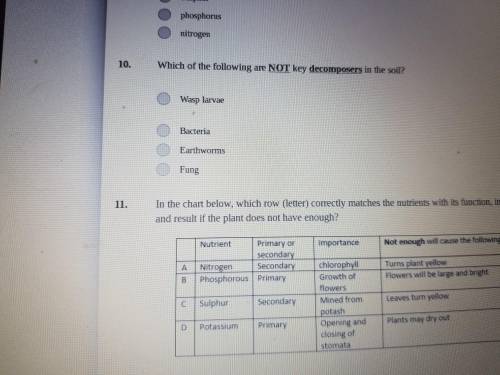
Answer please no links
...

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2

Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 1
You know the right answer?
Questions

History, 31.08.2021 03:50


Biology, 31.08.2021 03:50


Arts, 31.08.2021 03:50

English, 31.08.2021 03:50

Mathematics, 31.08.2021 03:50

Mathematics, 31.08.2021 03:50


History, 31.08.2021 03:50




Mathematics, 31.08.2021 03:50

Business, 31.08.2021 03:50

Geography, 31.08.2021 03:50

Mathematics, 31.08.2021 03:50


Social Studies, 31.08.2021 03:50

English, 31.08.2021 03:50