A chemist mixed two substances
together: a blue powder with no smell
and a colorless liquid...

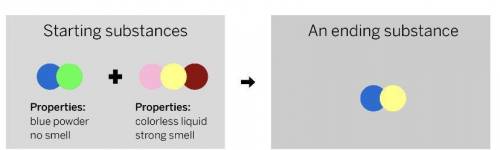
A chemist mixed two substances
together: a blue powder with no smell
and a colorless liquid with a strong
smell. Their repeating groups of atoms
are shown above on the left. After they
were mixed, the chemist analyzed the
results and found two substances. One
ending substance had the repeating
group of atoms shown above on the
right.
Is the ending substance the same
substance as the blue powder? What
happened to the atoms of the starting
substances when the ending
substances formed? Be sure to explain
your answers to both of these

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical solutions?
Answers: 1

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3
You know the right answer?
Questions




Arts, 13.12.2021 22:40


Mathematics, 13.12.2021 22:40

Mathematics, 13.12.2021 22:40

Mathematics, 13.12.2021 22:40

Mathematics, 13.12.2021 22:40

Mathematics, 13.12.2021 22:40

Mathematics, 13.12.2021 22:40

Chemistry, 13.12.2021 22:40

Social Studies, 13.12.2021 22:40


French, 13.12.2021 22:40

Chemistry, 13.12.2021 22:40

Mathematics, 13.12.2021 22:40

Computers and Technology, 13.12.2021 22:40





