
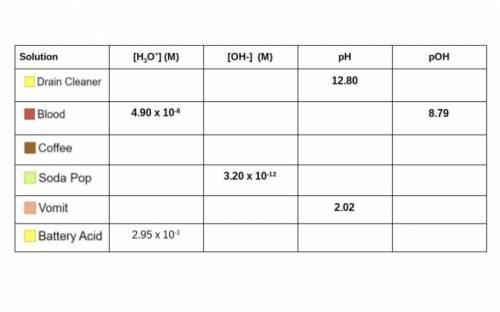
Use your knowledge of Molarity, pH/pOH, and the concentrations of ions to complete the table below. Some of
the work can be checked using the simulation. I have included a handy flow chart below the table with
common conversions when dealing with acids and bases. You will need to know how to do these calculations
for the unit test! (Picture attached)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1
You know the right answer?
Use your knowledge of Molarity, pH/pOH, and the concentrations of ions to complete the table below....
Questions


Social Studies, 20.02.2020 01:49

Mathematics, 20.02.2020 01:49


Mathematics, 20.02.2020 01:49



Social Studies, 20.02.2020 01:49



Mathematics, 20.02.2020 01:49

Chemistry, 20.02.2020 01:49

Mathematics, 20.02.2020 01:49


Social Studies, 20.02.2020 01:49



Mathematics, 20.02.2020 01:50

Mathematics, 20.02.2020 01:50

Mathematics, 20.02.2020 01:50



