Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:50
Nitrogen dioxide reacts with water to form nitric acid and nitrogen monoxide according to the equation: 3no2(g)+h2o(l)→2hno3(l)+no(g) part a suppose that 4.2 mol no2 and 0.50 mol h2o combine and react completely. which reactant is in excess? express your answer as a chemical formula. nothing
Answers: 1


Chemistry, 22.06.2019 06:00
How much would the freezing point of water decrease if 4 mol of sugar were added to 1 kg of water(k=1.86 c/mol/kg for water and i=1 for sugar
Answers: 1

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1
You know the right answer?
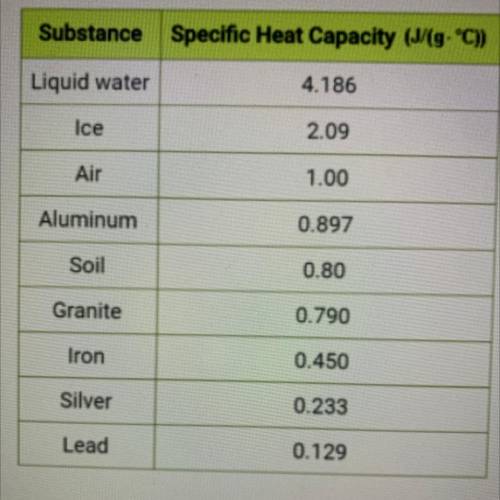
Use the specific heat value from the table at left to calculate the amount
of energy in Joules req...
Questions

English, 16.10.2019 00:30






History, 16.10.2019 00:30



English, 16.10.2019 00:30

Mathematics, 16.10.2019 00:30

Social Studies, 16.10.2019 00:30





Social Studies, 16.10.2019 00:30



Mathematics, 16.10.2019 00:30