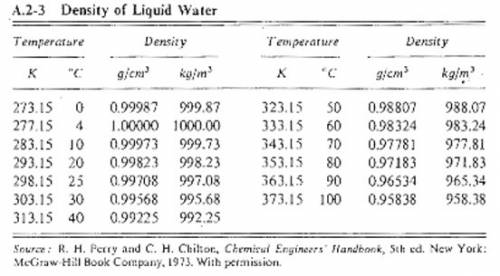
Suppose you were calibrating a 100.0 ml volumetric flask using distilled water. the flask temperature was at 20°c, and you assumed that the distilled water was as well. however, you later discover that the actual water temperature was 11°c instead. how is the mass of the 100.0 ml of distilled water you measured at 11°c different from the mass of 100.0 ml of distilled water at 20°c?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Which of the following compounds does not contain molecules? question 2 options: co2 h2 nacl h2o
Answers: 1

Chemistry, 22.06.2019 05:30
Which other elements contain the same number of outer electrons as sodium
Answers: 3

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 1
You know the right answer?
Suppose you were calibrating a 100.0 ml volumetric flask using distilled water. the flask temperatur...
Questions

Mathematics, 17.09.2020 08:01

English, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

Biology, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

English, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

Chemistry, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

English, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

English, 17.09.2020 08:01

Chemistry, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01

Biology, 17.09.2020 08:01

Mathematics, 17.09.2020 08:01