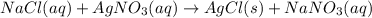Which describes a precipitate in a chemical reaction?
a. a gaseous reactant that forms aqueo...

Chemistry, 02.09.2019 09:10 calistaallen6655
Which describes a precipitate in a chemical reaction?
a. a gaseous reactant that forms aqueous products
b. a solid reactant that forms aqueous products
c. a gaseous product formed from aqueous reactants
d. a solid product formed from aqueous reactants

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3

Chemistry, 22.06.2019 17:30
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2
You know the right answer?
Questions


History, 18.10.2019 12:50




History, 18.10.2019 12:50



English, 18.10.2019 12:50

Social Studies, 18.10.2019 12:50

History, 18.10.2019 12:50

History, 18.10.2019 12:50

History, 18.10.2019 12:50


Mathematics, 18.10.2019 12:50




Mathematics, 18.10.2019 12:50

Mathematics, 18.10.2019 12:50