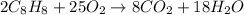Is this equation completely balanced? 2c8h8 + 25o2 8co2 + 18h2o
a. no, because the number of...

Chemistry, 11.10.2019 02:30 lilbrown6369
Is this equation completely balanced? 2c8h8 + 25o2 8co2 + 18h2o
a. no, because the number of carbon, hydrogen & oxygen atoms on both sides of the equation are not equal.
b. no, because the number of oxygen atoms on both sides of the equation are not equal.
c. yes, because all of the coefficients are in their lowest whole number ratio.
d. no, because the number of hydrogen atoms on both sides of the equation are not equal.
e. yes, because the number of products is equal to the number of reactants.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 00:30
Jessica is traveling from miami, florida, to chicago, illinois. using the map, tell one way the land will change during the second half of her trip.
Answers: 1


Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
Questions


English, 15.04.2020 07:57


Mathematics, 15.04.2020 07:57


Business, 15.04.2020 07:57


Mathematics, 15.04.2020 07:57

Social Studies, 15.04.2020 07:57

Social Studies, 15.04.2020 07:58

Biology, 15.04.2020 07:58



History, 15.04.2020 07:58



Biology, 15.04.2020 07:58

English, 15.04.2020 07:58