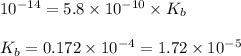Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1


Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2
You know the right answer?
The acid dissociation constant for boric acid, h3bo3, is 5.8 x 10-10. calculate the dissociation con...
Questions


Social Studies, 29.08.2019 16:30


Mathematics, 29.08.2019 16:30

Mathematics, 29.08.2019 16:30







English, 29.08.2019 16:30

History, 29.08.2019 16:30



Mathematics, 29.08.2019 16:30

Biology, 29.08.2019 16:30



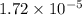
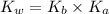
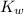 = Ionic product of water =
= Ionic product of water = 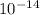
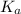 = Acid dissociation constant =
= Acid dissociation constant = 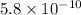
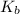 Base dissociation constant = ?
Base dissociation constant = ?