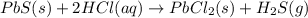Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Find the protons, electrons and neutrons for strontium with a mass of 83
Answers: 1


Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1
You know the right answer?
Solid lead(ii) sulfide reacts with aqueous hydrochloric acid to form solid lead(ii) chloride and dih...
Questions

Chemistry, 07.01.2021 23:20

Biology, 07.01.2021 23:20

Mathematics, 07.01.2021 23:20

Mathematics, 07.01.2021 23:20


Mathematics, 07.01.2021 23:20


Mathematics, 07.01.2021 23:20

History, 07.01.2021 23:20


English, 07.01.2021 23:20

English, 07.01.2021 23:20


Mathematics, 07.01.2021 23:20


Chemistry, 07.01.2021 23:20

Mathematics, 07.01.2021 23:20

Mathematics, 07.01.2021 23:20

Mathematics, 07.01.2021 23:20

Mathematics, 07.01.2021 23:20