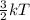Chemistry, 16.09.2019 21:30 younngchy92
If the temperature of a reaction is increased, the reaction proceeds at a much quicker rate because the

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Reaction rate depends on how many molecules are coming into contact with each other with enough energy to react. increasing the temperature of the reactants will increase -
Answers: 3

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 12:40
Consider the directing effects of the substituents on salicylamide and predict the possible structures of the iodination products. which do you think will be the major product?
Answers: 1
You know the right answer?
If the temperature of a reaction is increased, the reaction proceeds at a much quicker rate because...
Questions


Biology, 30.07.2020 01:01

Mathematics, 30.07.2020 01:01


English, 30.07.2020 01:01



Mathematics, 30.07.2020 01:01

Mathematics, 30.07.2020 01:01



Mathematics, 30.07.2020 01:01

Mathematics, 30.07.2020 01:01


Mathematics, 30.07.2020 01:01


Mathematics, 30.07.2020 01:01


Mathematics, 30.07.2020 01:01