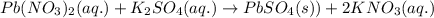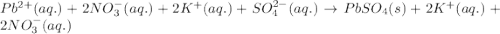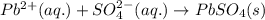Chemistry, 28.01.2020 08:31 Jazmineboo7709
Net ionic equation to show the reaction of aqueous lead(ii) nitrate with aqueous potassium sulfate to form solid lead(ii) sulfate and aqueous potassium nitrate.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 09:00
Avogradoa number was calculated by determining the number of atoms in?
Answers: 1
You know the right answer?
Net ionic equation to show the reaction of aqueous lead(ii) nitrate with aqueous potassium sulfate t...
Questions


Chemistry, 04.08.2019 07:20

Chemistry, 04.08.2019 07:20

English, 04.08.2019 07:20


Mathematics, 04.08.2019 07:20



Biology, 04.08.2019 07:20

Social Studies, 04.08.2019 07:20










Mathematics, 04.08.2019 07:20