

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 16:00
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1

Chemistry, 23.06.2019 04:10
Two solids are mixed in a flask and stirred. after a few minutes, the flask becomes cold. which of the following best describes this reaction? a. an exothermic reaction b. a combustion reaction c. an endothermic reaction d. a decomposition reaction
Answers: 1

Chemistry, 23.06.2019 05:30
What is the morality of 2.50 l of solution that contains 1.85 mol of anhydrous sodium tetraborate?
Answers: 1
You know the right answer?
Non-ferromagnetic materials tend to have electrons in each orbital...
Questions

Health, 13.10.2019 16:30

Mathematics, 13.10.2019 16:30

Computers and Technology, 13.10.2019 16:30



English, 13.10.2019 16:30


Chemistry, 13.10.2019 16:30

Mathematics, 13.10.2019 16:30


Physics, 13.10.2019 16:30

Mathematics, 13.10.2019 16:30

Mathematics, 13.10.2019 16:30

Chemistry, 13.10.2019 16:30


Mathematics, 13.10.2019 16:30

Mathematics, 13.10.2019 16:30



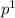 i.e. only one unpaired electron in the p- orbital. Now, consider iron its atomic number is 26 and its configuration is 3
i.e. only one unpaired electron in the p- orbital. Now, consider iron its atomic number is 26 and its configuration is 3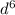 . It has more number of unpaired electrons. Thus, neon is non-magnetic, aluminum is also non-magnetic but iron is magnetic.
. It has more number of unpaired electrons. Thus, neon is non-magnetic, aluminum is also non-magnetic but iron is magnetic.

