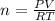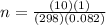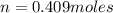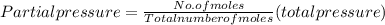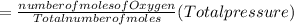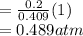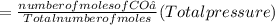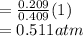Chemistry, 13.10.2019 23:30 jasminelynn135owmyj1
A10 liter flask at 298 k contains a gaseous mixture of o2 and co2 at 1 atmosphere. which statement is true for the partial pressures of o2 and co2 if 0.2 mole of o2 is present in the flask? (given the universal gas constant r = 0.082 l∙atm/k∙mol)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:40
For a patient with the following pes statement and interventions, which would be the most appropriate monitoring and evaluating data? pes statement: inadequate calcium intake related to food and nutrition related knowledge deficit as evidenced by statements that the only dietary source of calcium is milk and she believes that she is lactose intolerant. patient’s nutrition prescription is for a diet providing 1200 mg calcium per day. patient was provided with in-depth nutrition education on alternative dietary and supplement sources of calcium. a. calcium intake (at subsequent visit) b. knowledge assessment by asking patient to identify food sources from menus and shopping list (at the end of the current visit) c. serum calcium (at next visit) d. both a and b e. both a and c
Answers: 2

Chemistry, 22.06.2019 05:30
What happens to the atomic radius when an elctron is lost
Answers: 1


Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1
You know the right answer?
A10 liter flask at 298 k contains a gaseous mixture of o2 and co2 at 1 atmosphere. which statement i...
Questions


History, 22.07.2019 00:32

History, 22.07.2019 00:32

Mathematics, 22.07.2019 00:32

Mathematics, 22.07.2019 00:32


Chemistry, 22.07.2019 00:32

Mathematics, 22.07.2019 00:32

Mathematics, 22.07.2019 00:32





Mathematics, 22.07.2019 00:32


English, 22.07.2019 00:32


Spanish, 22.07.2019 00:32