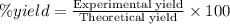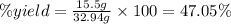One method to produce nitrogen in the lab is to react ammonia with copper (ii) oxide: nh3(
g....

Chemistry, 30.09.2019 12:00 Juliette9525
One method to produce nitrogen in the lab is to react ammonia with copper (ii) oxide: nh3(
g. + cuo(s) cu(s) + h2o(l) + n2(
g. after using 40.0 grams of nh3, 15.5 grams of n2 are produced. what is the percent yield of nitrogen in the reaction?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Find the protons, electrons and neutrons for strontium with a mass of 83
Answers: 1


Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

You know the right answer?
Questions

Physics, 19.07.2019 17:00



Computers and Technology, 19.07.2019 17:00



History, 19.07.2019 17:00

Mathematics, 19.07.2019 17:00

Computers and Technology, 19.07.2019 17:00

Social Studies, 19.07.2019 17:00


Biology, 19.07.2019 17:00

History, 19.07.2019 17:00

Biology, 19.07.2019 17:00

Social Studies, 19.07.2019 17:00

History, 19.07.2019 17:00


History, 19.07.2019 17:00


Chemistry, 19.07.2019 17:00

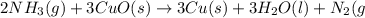
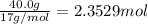
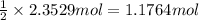 of nitrogen gas
of nitrogen gas