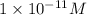Answer : The correct option is, (C) 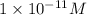
Explanation : Given,
pH = 11
pH : It is defined as the negative logarithm of hydrogen ion cocnetration.
Mathematically it is represented as :
![pH=-\log [H^+]](/tpl/images/0332/3042/37e81.png)
Now put the value of pH in this expression, we get the hydrogen ion concentration.
![11=-\log [H^+]](/tpl/images/0332/3042/95b83.png)
![[H^+]=1.0\times 10^{-11}M](/tpl/images/0332/3042/4ccae.png)
Therefore, the hydrogen ion concentration is