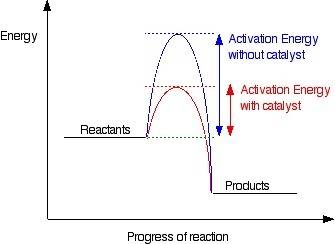
How do catalysts increase the rate of a chemical reaction?
they lower the activation energy.<...

Chemistry, 23.01.2020 10:31 christianmason9423
How do catalysts increase the rate of a chemical reaction?
they lower the activation energy.
they lower the collision energy.
they increase the collision energy.
they increase the temperature.
they increase the concentration of reactants.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


You know the right answer?
Questions

Biology, 21.09.2019 00:10