
Chemistry, 03.11.2019 01:31 jjjjjjgegi3088
State and explain the effect of increasing the pressure on the yield of ammonia in this reaction (decomposition of ammonium carbonate).
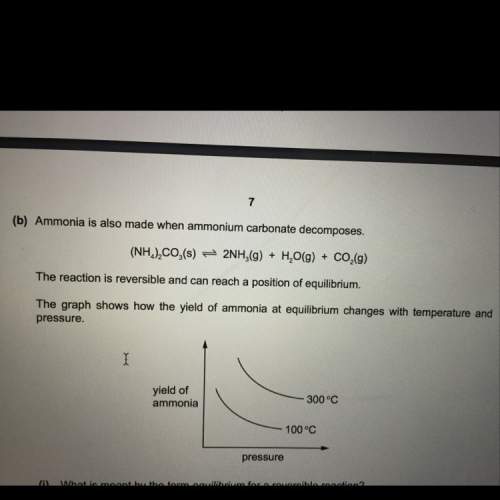

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2

Chemistry, 23.06.2019 00:30
Nuclear decay is the spontaneous decay of one element into a. an x-ray b. a ray of light c. another element
Answers: 1

Chemistry, 23.06.2019 03:30
Name 3 types of energy you see being used as you look around a classroom
Answers: 1
You know the right answer?
State and explain the effect of increasing the pressure on the yield of ammonia in this reaction (de...
Questions


English, 09.12.2020 20:30

Mathematics, 09.12.2020 20:30

English, 09.12.2020 20:30



Mathematics, 09.12.2020 20:30

Mathematics, 09.12.2020 20:30

Mathematics, 09.12.2020 20:30


Mathematics, 09.12.2020 20:30

SAT, 09.12.2020 20:30

Social Studies, 09.12.2020 20:30

Health, 09.12.2020 20:30








