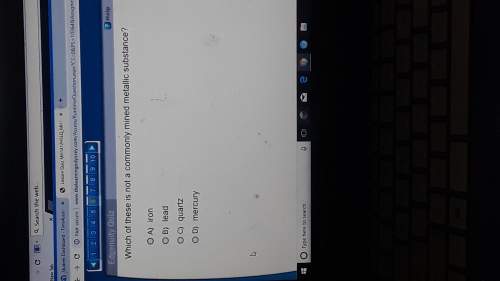
Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Water molecules have a strong attraction to each other because of hydrogen bonding, allowing water to move against gravity up a plant's stem through capillary action. true false
Answers: 2

Chemistry, 22.06.2019 02:40
For a patient with the following pes statement and interventions, which would be the most appropriate monitoring and evaluating data? pes statement: inadequate calcium intake related to food and nutrition related knowledge deficit as evidenced by statements that the only dietary source of calcium is milk and she believes that she is lactose intolerant. patient’s nutrition prescription is for a diet providing 1200 mg calcium per day. patient was provided with in-depth nutrition education on alternative dietary and supplement sources of calcium. a. calcium intake (at subsequent visit) b. knowledge assessment by asking patient to identify food sources from menus and shopping list (at the end of the current visit) c. serum calcium (at next visit) d. both a and b e. both a and c
Answers: 2

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 07:30
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1
You know the right answer?
How can you cause a physical change...
Questions

Chemistry, 06.07.2019 21:00


Mathematics, 06.07.2019 21:00


Social Studies, 06.07.2019 21:00



History, 06.07.2019 21:00

Business, 06.07.2019 21:00


Computers and Technology, 06.07.2019 21:00

Biology, 06.07.2019 21:00


Spanish, 06.07.2019 21:00


Mathematics, 06.07.2019 21:00

History, 06.07.2019 21:00

Mathematics, 06.07.2019 21:00

Geography, 06.07.2019 21:00

Mathematics, 06.07.2019 21:00