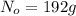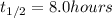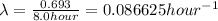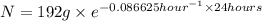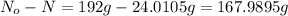Chemistry, 15.11.2019 17:31 KitKat22Rose9
Acertain radioisotope has a half-life of 8.0 hours; how much of a 192 g sample of this radioisotope would decay after a full day?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:00
One significant difference between an ionic bond, where electrons are taken from one atom and added to another atom, and a covalent or metallic bond, where electrons are shared is
Answers: 2

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

Chemistry, 22.06.2019 20:40
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1
You know the right answer?
Acertain radioisotope has a half-life of 8.0 hours; how much of a 192 g sample of this radioisotope...
Questions


Biology, 08.07.2019 04:10

Chemistry, 08.07.2019 04:10

Chemistry, 08.07.2019 04:10

History, 08.07.2019 04:10


History, 08.07.2019 04:10


Chemistry, 08.07.2019 04:20




Biology, 08.07.2019 04:20







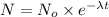
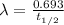
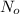 = initial amount of radioisotope
= initial amount of radioisotope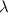 = Decay Constant
= Decay Constant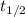 = Half life of the radioisotope
= Half life of the radioisotope