
Avogadro’s law and charles’s law describe a proportionality of the volume of a gas when the pressure is constant. which describes the proportionality that allows these laws to be combined when describing a gas?
volume is directly proportional to temperature (avogadro’s law) and to moles (charles’s law).
volume is inversely proportional to temperature (avogadro’s law) and to moles (charles’s law).
volume is directly proportional to moles (avogadro’s law) and to temperature (charles’s law).
volume is inversely proportional to moles (avogadro’s law) and to temperature (charles’s law).

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 22.06.2019 22:30
Akno3 solution containing 51 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 3

Chemistry, 23.06.2019 02:30
What role does weathering have in shaping earth’s surface? a) it allows sediments to fall out of a medium. b) it sediments settle on a new surface. c) it breaks down older material into sediments. d) it transports sediments to a different location. will give brainliest, answer quickly.
Answers: 2
You know the right answer?
Avogadro’s law and charles’s law describe a proportionality of the volume of a gas when the pressure...
Questions


History, 10.04.2020 06:40

Geography, 10.04.2020 06:40



Mathematics, 10.04.2020 06:40

Mathematics, 10.04.2020 06:40


Social Studies, 10.04.2020 06:40

English, 10.04.2020 06:40




History, 10.04.2020 06:41

Advanced Placement (AP), 10.04.2020 06:41


Mathematics, 10.04.2020 06:41

Mathematics, 10.04.2020 06:41

Mathematics, 10.04.2020 06:41

Mathematics, 10.04.2020 06:42

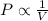 (At constant temperature and number of moles)
(At constant temperature and number of moles)
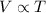 (At constant pressure and number of moles)
(At constant pressure and number of moles)
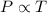 (At constant volume and number of moles)
(At constant volume and number of moles)
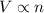 (At constant temperature and pressure)
(At constant temperature and pressure)

