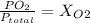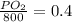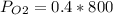Chemistry, 13.11.2019 09:31 blueflu5120
Amixture of gases with a pressure of 800.0 mm hg contains 60% nitrogen and 40% oxygen by volume. what is the partial pressure of oxygen in this mixture?
a. 140.0 mm hg
b. 320.0 mm hg
c. 373.0 mm hg
d. 480.0 mm hg

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3


Chemistry, 23.06.2019 03:00
Analyze the reaction to determine whether the reaction is exothermic or endothermic. explain your reasoning.
Answers: 1

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1
You know the right answer?
Amixture of gases with a pressure of 800.0 mm hg contains 60% nitrogen and 40% oxygen by volume. wha...
Questions


Mathematics, 17.02.2021 17:20

History, 17.02.2021 17:20

Mathematics, 17.02.2021 17:20

Mathematics, 17.02.2021 17:20


Mathematics, 17.02.2021 17:20

Mathematics, 17.02.2021 17:20

English, 17.02.2021 17:20



Mathematics, 17.02.2021 17:20


Social Studies, 17.02.2021 17:20




Mathematics, 17.02.2021 17:20

History, 17.02.2021 17:20

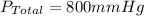
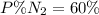
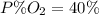
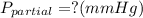
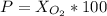
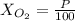
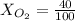
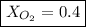
 :
: