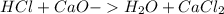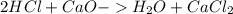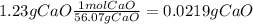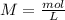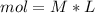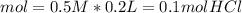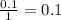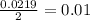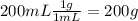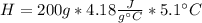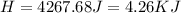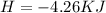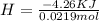During an experiment, a student adds 1.23 g of cao to 200.0 ml of 0.500 m hcl. the student observes a temperature increase of 5.10 °c. assuming the solution\'s final volume is 200.0 ml, the density if 1.00 g/ml, and the heat capacity is 4.184 j/(g·°c, calculate the heat of the reaction, ? hrxn.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

You know the right answer?
During an experiment, a student adds 1.23 g of cao to 200.0 ml of 0.500 m hcl. the student observes...
Questions


Mathematics, 19.06.2020 03:57






Mathematics, 19.06.2020 03:57



Mathematics, 19.06.2020 03:57


Mathematics, 19.06.2020 03:57

Mathematics, 19.06.2020 03:57

Mathematics, 19.06.2020 03:57

Mathematics, 19.06.2020 03:57


History, 19.06.2020 03:57

Mathematics, 19.06.2020 03:57

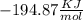
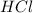 and
and 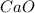 will produce
will produce 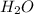 and
and 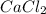 plus energy, because is an exothermic reaction. The first step is step up the reaction:
plus energy, because is an exothermic reaction. The first step is step up the reaction: