
Chemistry, 27.08.2019 02:50 quinteroalex2001
Which reaction is balanced correctly? c + o2 → 2co2 ch4 + 4o2 → co2 + 2h2o c3h8 + 5o2 → 3co2 + 4h2o c6h12o6 + 3o2 → 6co2 + 6h2o

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:20
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2(g)→2alcl3(s) what is the maximum mass of aluminum chloride that can be formed when reacting 32.0 g of aluminum with 37.0 g of chlorine? express your answer to three significant figures and include the appropriate units.
Answers: 2

Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3
You know the right answer?
Which reaction is balanced correctly? c + o2 → 2co2 ch4 + 4o2 → co2 + 2h2o c3h8 + 5o2 → 3co2 + 4h2o...
Questions

Mathematics, 28.06.2019 00:10

Biology, 28.06.2019 00:10


Mathematics, 28.06.2019 00:10



Biology, 28.06.2019 00:10


Mathematics, 28.06.2019 00:10

Social Studies, 28.06.2019 00:10

Social Studies, 28.06.2019 00:10









Mathematics, 28.06.2019 00:10

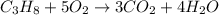 is balanced correctly
is balanced correctly

