
Chemistry, 05.10.2019 18:30 sportygirlscand
Calculate the percent ionization of nitrous acid in a solution that is 0.139 m in nitrous acid. the acid dissociation constant of nitrous acid is 4.50 ⋅ 10-4.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 22.06.2019 21:00
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1

Chemistry, 22.06.2019 21:40
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1
You know the right answer?
Calculate the percent ionization of nitrous acid in a solution that is 0.139 m in nitrous acid. the...
Questions

Mathematics, 25.06.2019 17:00

Mathematics, 25.06.2019 17:00

Mathematics, 25.06.2019 17:00

Mathematics, 25.06.2019 17:00


Mathematics, 25.06.2019 17:00


History, 25.06.2019 17:00

History, 25.06.2019 17:00

Biology, 25.06.2019 17:00





Mathematics, 25.06.2019 17:00



Mathematics, 25.06.2019 17:00


Mathematics, 25.06.2019 17:00

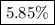 .
.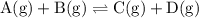
![{\text{K}}=\frac{{\left[{\text{D}}\right]\left[{\text{C}}\right]}}{{\left[{\text{A}}\right]\left[ {\text{B}}\right]}}](/tpl/images/0290/4975/92f42.png)
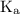 and equilibrium constant for the dissociation of base is known as
and equilibrium constant for the dissociation of base is known as 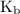 .
.![\% {\text{ ionization}}=\frac{{\left[ {{{\text{H}}_{\text{3}}}{{\text{O}}^{\text{ + }}}}\right]}}{{\left[{{\text{HA}}}\right]}}\times100](/tpl/images/0290/4975/5479a.png)
![\left[{{{\text{H}}_{\text{3}}}{{\text{O}}^{\text{+}}}}\right]](/tpl/images/0290/4975/53aa9.png) is the concentration of hydronium ion and
is the concentration of hydronium ion and ![\left[{{\text{HA}}}\right]](/tpl/images/0290/4975/88419.png) is the concentration of acid.
is the concentration of acid.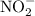 and hydronium ion.
and hydronium ion.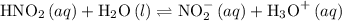
![\% {\text{ ionization}}=\frac{{\left[{{{\text{H}}_{\text{3}}}{{\text{O}}^{\text{+}}}}\right]}}{{\left[{{\text{HN}}{{\text{O}}_2}}\right]}}\times100](/tpl/images/0290/4975/29d81.png) …… (2)
…… (2)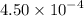 .
.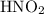 becomes 0139-x at equilibrium. The concentration of
becomes 0139-x at equilibrium. The concentration of 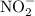 and
and 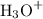 ion becomes x at equilibrium.
ion becomes x at equilibrium.![\left[{{\text{NO}}_2^-}\right]](/tpl/images/0290/4975/bb69c.png) and 0.139-x for
and 0.139-x for 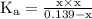 …… (3)
…… (3)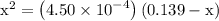
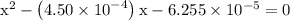
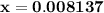
![\left[{{\text{HN}}{{\text{O}}_2}}\right]](/tpl/images/0290/4975/958ed.png) in equation (2).
in equation (2).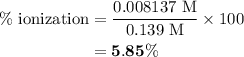
 (aq):
(aq): 

