

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1


Chemistry, 23.06.2019 03:30
How many grams of sodium chloride are in 250ml of a 2.5m naci solution
Answers: 1
You know the right answer?
How many covalent bonds does carbon form if each of its unpaired electrons participate in one bond?...
Questions

English, 09.11.2020 17:50

Mathematics, 09.11.2020 17:50


History, 09.11.2020 17:50

Mathematics, 09.11.2020 17:50

Social Studies, 09.11.2020 17:50

Mathematics, 09.11.2020 17:50


Mathematics, 09.11.2020 17:50

Mathematics, 09.11.2020 17:50


Chemistry, 09.11.2020 17:50


Mathematics, 09.11.2020 17:50


Health, 09.11.2020 17:50

Mathematics, 09.11.2020 17:50


Mathematics, 09.11.2020 17:50

Biology, 09.11.2020 17:50

![\boxed{ \ _{6}C = 1s^2 \ 2s^2 \ 2p^2 \ or \ [He] \ 2s^2 \ 2p^2 \ }](/tpl/images/0307/4498/64492.png)
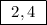 The electrons are distributed in the 1st and 2nd orbit. K shell (n = 1) = 2 electrons.
L shell (n = 2) = 4 electrons.
The electrons are distributed in the 1st and 2nd orbit. K shell (n = 1) = 2 electrons.
L shell (n = 2) = 4 electrons.

