
Chemistry, 28.01.2020 21:58 alaina3792
When a person has excess stomach acid, he or she may ingest some sodium bicarbonate. the rationale for doing this is that the sodium bicarbonate is:
a base and will neutralize the excess acid
an acid and will weaken the excess acid
neutral and will dilute the excess acid
it will make the person thirsty; they will drink water and dilute the excess acid.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

You know the right answer?
When a person has excess stomach acid, he or she may ingest some sodium bicarbonate. the rationale f...
Questions


Computers and Technology, 09.10.2019 04:40

English, 09.10.2019 04:40

English, 09.10.2019 04:50

Mathematics, 09.10.2019 04:50


Geography, 09.10.2019 04:50


Biology, 09.10.2019 04:50

Mathematics, 09.10.2019 04:50


History, 09.10.2019 04:50

Biology, 09.10.2019 04:50



Mathematics, 09.10.2019 04:50

Chemistry, 09.10.2019 04:50


Health, 09.10.2019 04:50

English, 09.10.2019 04:50

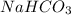 . It is basic in nature as it dissociates to give sodium and bicarbonate ions.
. It is basic in nature as it dissociates to give sodium and bicarbonate ions. 

