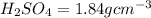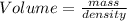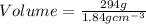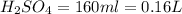2. sulfur dioxide gas (so2) reacts with excess oxygen gas (o2) and excess liquid water (h2o) to form liquid sulfuric acid (h2so4) in the unbalanced equation below: so2 + o2 + h2o h2so4 in the laboratory, a chemist carries out this reaction at stp with 67.2 l of sulfur dioxide (so2). how many liters of h2so4 did the chemist produce? 1 mole of any gas = 22.4 l of that same gas at stp • part a: write a balanced equation for the reaction. • part b: calculate the number of liters of h2so4 produced.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2


Chemistry, 22.06.2019 19:00
Suppose that a certain fortunate person has a net worth of $71.0 billion ($7.10×1010). if her stock has a good year and gains $3.20 billion (3.20×109) in value, what is her new net worth?
Answers: 3
You know the right answer?
2. sulfur dioxide gas (so2) reacts with excess oxygen gas (o2) and excess liquid water (h2o) to form...
Questions




Mathematics, 14.04.2020 20:12


History, 14.04.2020 20:12

Chemistry, 14.04.2020 20:12

Mathematics, 14.04.2020 20:12

English, 14.04.2020 20:12


History, 14.04.2020 20:13



Mathematics, 14.04.2020 20:13



History, 14.04.2020 20:13



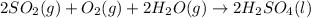
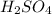 =0.16L
=0.16L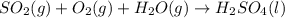
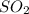 occupies 22.4 L at STP
occupies 22.4 L at STP 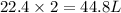 and produce 2 moles of
and produce 2 moles of 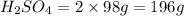
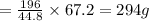 of
of