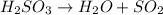Chemistry, 06.10.2019 22:50 kristy2958
Sulfurous acid, h2so3, breaks down into water (h2o) and sulfur dioxide (so2). if only one molecule of sulfurous acid was involved in a chemical reaction, how many sulfur (s) atoms would be in the products?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1
You know the right answer?
Sulfurous acid, h2so3, breaks down into water (h2o) and sulfur dioxide (so2). if only one molecule o...
Questions













Business, 23.06.2019 09:30

Computers and Technology, 23.06.2019 09:30

Spanish, 23.06.2019 09:30



Mathematics, 23.06.2019 09:30


Mathematics, 23.06.2019 09:30